Health
FDA Memo Reveals Link Between Child Deaths and COVID-19 Vaccines
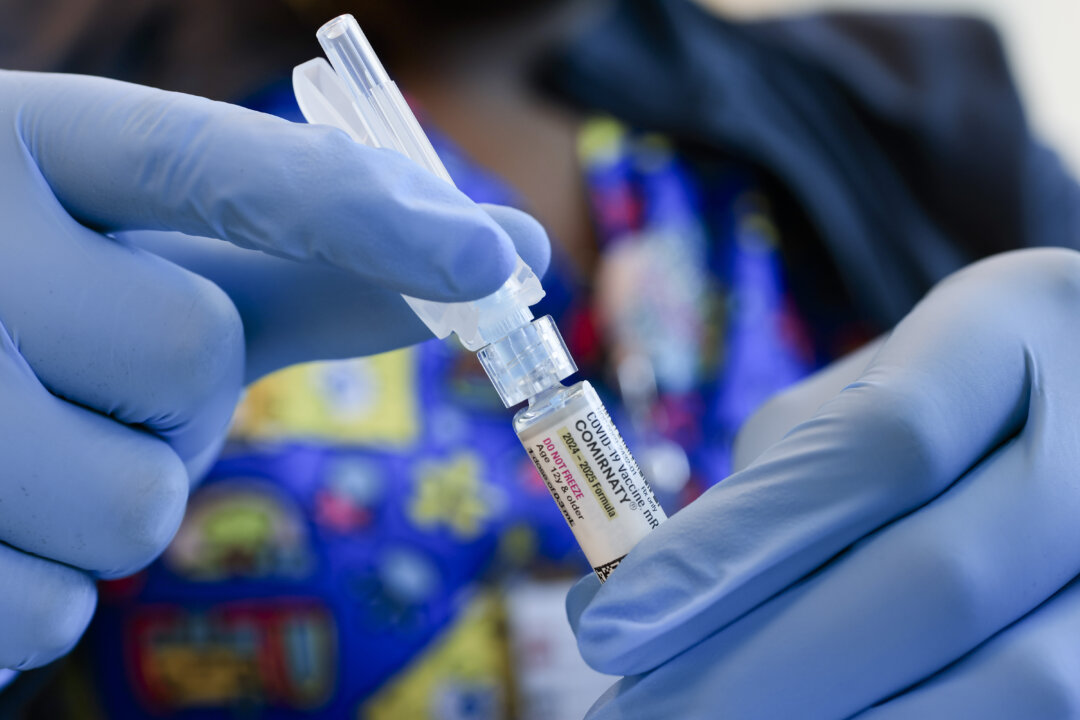
An internal memo from the Food and Drug Administration (FDA) has disclosed that agency scientists have identified at least 10 child deaths they believe are associated with COVID-19 vaccination. This revelation, reported by The Epoch Times, marks a significant acknowledgment within the FDA regarding the potential risks of vaccines for children.
The memo, authored by Dr. Vinay Prasad, who leads the FDA’s Center for Biologics Evaluation and Research (CBER), highlights findings from an evaluation conducted by the Office of Biostatistics and Pharmacovigilance. This analysis reviewed 96 death reports voluntarily submitted to the Vaccine Adverse Event Reporting System (VAERS) between 2021 and 2024. The staff concluded that at least ten of these cases met the criteria for a likely, probable, or possible causal link to the COVID-19 vaccines.
Insights from the FDA Analysis
Dr. Prasad’s memo emphasizes the importance of ongoing investigations into vaccine safety, particularly for vulnerable populations such as children. The internal review sought to determine the circumstances surrounding these deaths and assess whether the vaccines could be a contributing factor. The acknowledgment of these cases has sparked discussions about vaccine safety protocols and the need for transparent communication regarding potential risks.
According to the memo, the criteria used to establish a causal link were stringent. The evaluation process involved analyzing various factors, including the medical histories of the children and the timing of the deaths in relation to vaccination. The findings underscore the ongoing debate around vaccine safety, especially as vaccines have become a central component of public health strategies against COVID-19.
The Broader Implications
The implications of this memo are significant, particularly as governments and health organizations continue to encourage vaccination among children. The identification of potential risks has raised questions among parents, healthcare professionals, and policymakers regarding the balance between vaccination benefits and safety concerns.
While the FDA has historically emphasized the safety and efficacy of vaccines, this internal acknowledgment could lead to increased scrutiny of vaccination programs. Stakeholders may call for more extensive studies and data transparency to ensure public confidence in vaccination initiatives.
In light of these developments, health authorities are likely to face pressure to provide clearer guidelines and information on vaccine safety, particularly for younger populations. As the situation evolves, ongoing communication and research will be crucial in addressing public concerns and ensuring that vaccination remains a safe and effective tool in combating COVID-19.
As discussions continue, the FDA’s commitment to evaluating vaccine safety will be closely monitored by both the public and health professionals. The agency faces the challenge of balancing vaccine promotion with the need for transparency about potential risks, particularly in light of these new findings.
-

 Science4 weeks ago
Science4 weeks agoUniversity of Hawaiʻi Joins $25.6M AI Initiative to Monitor Disasters
-

 Lifestyle2 months ago
Lifestyle2 months agoToledo City League Announces Hall of Fame Inductees for 2024
-

 Business2 months ago
Business2 months agoDOJ Seizes $15 Billion in Bitcoin from Major Crypto Fraud Network
-

 Top Stories2 months ago
Top Stories2 months agoSharp Launches Five New Aquos QLED 4K Ultra HD Smart TVs
-

 Sports2 months ago
Sports2 months agoCeltics Coach Joe Mazzulla Dominates Local Media in Scrimmage
-

 Politics2 months ago
Politics2 months agoMutual Advisors LLC Increases Stake in SPDR Portfolio ETF
-

 Health2 months ago
Health2 months agoCommunity Unites for 7th Annual Walk to Raise Mental Health Awareness
-

 Science2 months ago
Science2 months agoWestern Executives Confront Harsh Realities of China’s Manufacturing Edge
-

 World2 months ago
World2 months agoINK Entertainment Launches Exclusive Sofia Pop-Up at Virgin Hotels
-

 Politics2 months ago
Politics2 months agoMajor Networks Reject Pentagon’s New Reporting Guidelines
-

 Science1 month ago
Science1 month agoAstronomers Discover Twin Cosmic Rings Dwarfing Galaxies
-

 Top Stories1 month ago
Top Stories1 month agoRandi Mahomes Launches Game Day Clothing Line with Chiefs